Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (30): 4831-4837.doi: 10.3969/j.issn.2095-4344.2014.30.012
Previous Articles Next Articles
Mechanical and biological properties of porous calcium phosphate cement/fibrin glue for bone regeneration in vitro
Dong Jing-jing1, Bi Long2, Li Na3, Guo Yu1, Xu Qiang1, Lei Wei2, Cui Geng4
- 1Lintong Aeromedical Evaluation and Training Center of Chinese Air Force, Xi’an 710600, Shaanxi Province, China
2Department of Orthopedics, Xijing Hospital, the Fourth Military Medical University, Xi’an 710032, Shaanxi Province, China
3Lintong Sanatorium of Lanzhou Military Command, Xi’an 710600, Shaanxi Province, China
4Department of Orthopedics, the 301 Hospital of Chinese PLA, Beijing 100039, Chin
-
Revised:2014-06-15Online:2014-07-16Published:2014-08-08 -
Contact:Cui Geng, Associate chief physician, Department of Orthopedics, the 301 Hospital of Chinese PLA, Beijing 100039, China -
About author:Dong Jing-jing, M.D., Attending physician, Lintong Aeromedical Evaluation and Training Center of Chinese Air Force, Xi’an 710600, Shaanxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81000790; Scientific New Star Plan of Beijing City, No. 2010-77
CLC Number:
Cite this article
Dong Jing-jing, Bi Long, Li Na, Guo Yu, Xu Qiang, Lei Wei, Cui Geng . Mechanical and biological properties of porous calcium phosphate cement/fibrin glue for bone regeneration in vitro[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(30): 4831-4837.
share this article
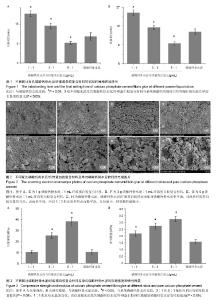
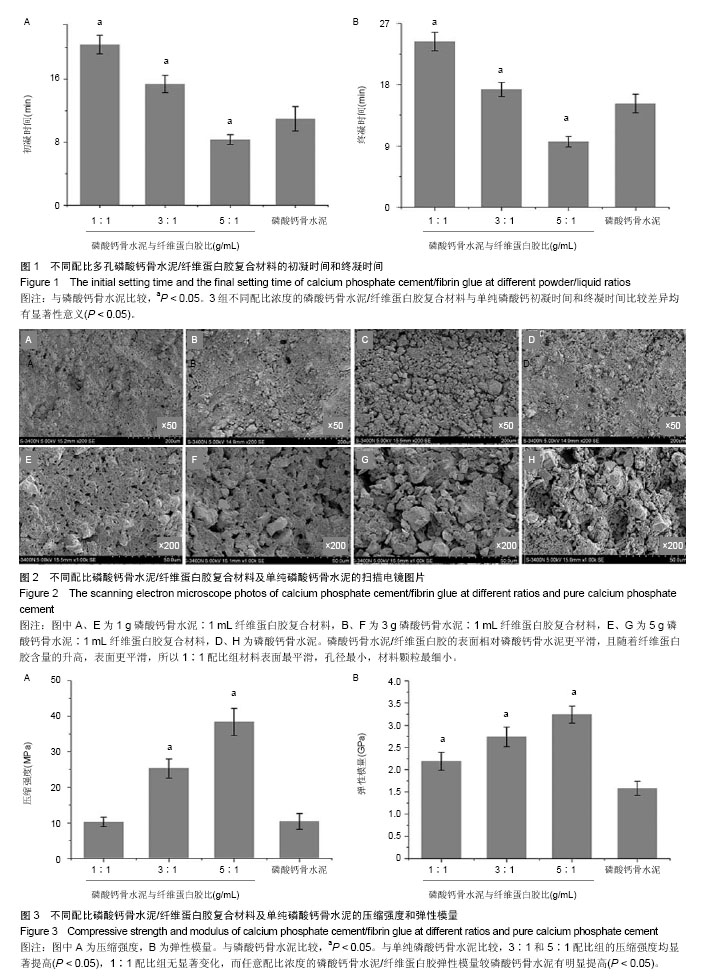
2.1 初始凝固时间和终末凝固时间的测定 磷酸钙骨水泥/纤维蛋白胶配比浓度为1∶1和3∶1时,初凝时间较单纯磷酸钙明显延长,且1∶1配比初凝时间更长,达到(20.41±1.21) min (P < 0.05);磷酸钙骨水泥/纤维蛋白胶配比浓度为5∶1时,初凝时间则较单纯磷酸钙缩短(P < 0.05);终凝时间也表现出相同的特点,1∶1配比组终凝时间最长,5∶1配比组最短;3组不同配比浓度的磷酸钙骨水泥/纤维蛋白胶复合材料与单纯磷酸钙初凝时间和终凝时间比较差异均有显著性意义(P < 0.05),见图1。 2.2 扫描电镜观察结果 任意配比浓度的磷酸钙骨水泥/纤维蛋白胶和单纯磷酸钙骨水泥的表面结构存在明显差异,磷酸钙骨水泥/纤维蛋白胶的表面相对磷酸钙骨水泥更平滑,且随着纤维蛋白胶含量的升高,表面更平滑,所以1∶1配比组材料表面最平滑,孔径最小,材料颗粒最细小;单纯磷酸钙骨水泥材料表面最粗糙,颗粒直径相差较大,有较大的孔洞结构(图2)。 2.3 体外生物力学检测结果 图3为各组材料在室温条件下充分干燥后测得的体外压缩强度和弹性模量。与单纯磷酸钙骨水泥比较,3∶1和5∶1配比组的压缩强度均显著提高(P < 0.05),1∶1配比组无显著变化,而任意配比浓度的磷酸钙骨水泥/纤维蛋白胶弹性模量较磷酸钙骨水泥有明显提高(P < 0.05),且5∶1配比组最高。"
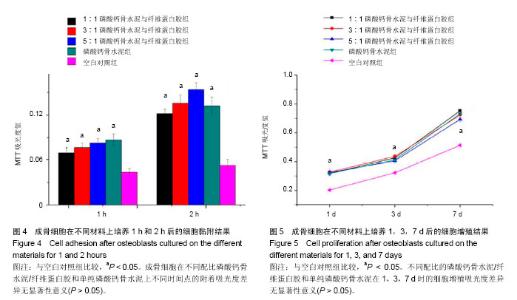
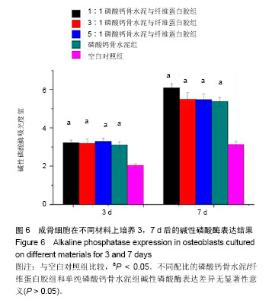
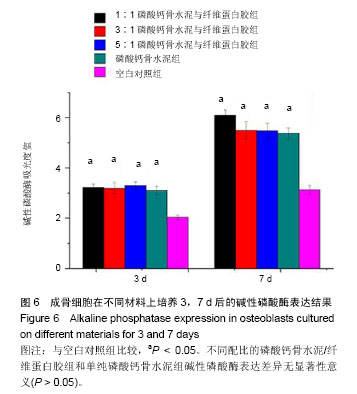
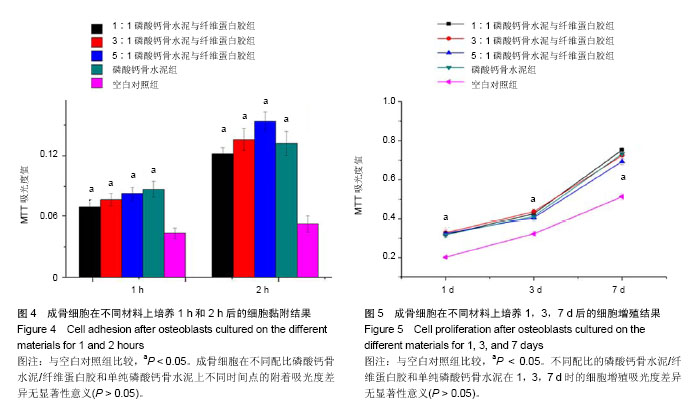
2.4 不同配比复合材料磷酸钙骨水泥/纤维蛋白胶对成骨细胞黏附的影响 如图4所示,成骨细胞在不同配比的磷酸钙骨水泥/纤维蛋白胶材料及单纯的磷酸钙骨水泥上培养黏附1 h和2 h后,其吸光度值明显高于空白对照组(P < 0.05),且所有组2 h较1 h的细胞黏附吸光度值高(P < 0.05),但不同配比磷酸钙骨水泥/纤维蛋白胶组和单纯的磷酸钙骨水泥在1 h和2 h时,成骨细胞附着的吸光度差异无显著性意义(P > 0.05)。 2.5 不同配比复合材料磷酸钙骨水泥/纤维蛋白胶对成骨细胞增殖的影响 如图5所示,成骨细胞在各组材料上分别培养1,3,7 d后,其细胞增殖的吸光度均明显高于空白对照组(P < 0.05),但不同配比的磷酸钙骨水泥/纤维蛋白胶组和单纯磷酸钙骨水泥组在1,3,7 d时的细胞增殖吸光度差异无显著性意义(P > 0.05)。"
| [1] Miyazaki M,Tsumura H,Wang JC,et al.An update on bone substitutes for spinal fusion.Eur Spine J.2009;18(6):783-799. |
| [1] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [2] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [3] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [4] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [5] | Chen Qiaoling, Bai Yiguang, Liu Kang, Lin Tao, Luo Xuwei. Osteoblast differentiation after conditional knockout of 3-phosphoinositide-dependent protein kinase-1 gene from bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3785-3789. |
| [6] | You Wulin, Huang Guicheng, Wang Jianwei. Osteogenic differentiation potential of microencapsulated transgenic bone marrow mesenchymal stem cells cocultured with osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3852-3857. |
| [7] | Wang Weiwei, Ou Zhixue, Zhang Xiaoyun, Li Shibin, Zhou Yi, Li Tong. Regulatory mechanism of exosomes in signal communication network of steroid induced avascular necrosis of the femoral head repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3056-3064. |
| [8] | Wang Jing, Wang Wanyuji, Zhang Yi, Ma Yaping, Wang Xin. Interleukin 6 involved in a series of reaction processes of osteogenesis and bone repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2945-2951. |
| [9] | Zhang Hongmei, Sun Xirao, Wang Chengyue. Effect of polymethyl methacrylate/mineralized collagen/Mg-Ca composite material on osteogenic differentiation of mouse preosteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2498-2503. |
| [10] | Yang Bingxuan, Jiang Tao, Jia Min, Li Peng, Liao Rongzhen, Li Zhao, Shao Min. Icariin promotes differentiation of mouse preosteoblasts via activating autophagy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 64-69. |
| [11] | Huang Tao, Cheng Zhijian, Jia Zhiqiang, Zhao Xiaoguang, Wang Lei, Zhai Wenjing, Zhou Yongxin. Mechanism by which miR-146a regulates osteogenic differentiation of adipose derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 70-75. |
| [12] | Feng Zhiguo, Sun Haibiao, Han Xiaoqiang. Regulation of proliferation, differentiation and apoptosis of bone-related cells by long-stranded non-coding RNA [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 112-118. |
| [13] | Wang Jing, Lang Xuemei, Wang Weiqun, Zhang Hanxiang, Zhang Yi, Wang Xin. Participation and regulatory mechanism of interleukin-1 during bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(36): 5851-5858. |
| [14] | Zhang Mingjin, Zhou Yanheng, Liu Dawei. Reopening the fused suture provides a new idea for maxillary expansion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4721-4727. |
| [15] | Li Ruozhen, Tian Yaping, Wen Ning. Biological effect of magnetic fields to promote bone responses to biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(28): 4510-4515. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 159
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 286
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||